Zenogen Pharma HSC-BANKER GMP grade
조혈줄기세포 동결보존용 GMP 등급 시약으로, 혈청 및 동물 유래 성분이 없는 Ready-to-use 제품입니다. DMSO/DEXTRAN 기반 기존 프로토콜과 동등 이상의 보존 효율을 제공합니다. JP, EU, US, PIC/S GMP 기준에 따라 제조되며 2–8℃에서 3년 안정성을 유지합니다.
✨AI 추천 연관 상품
AI가 분석한 이 상품과 연관된 추천 상품들을 확인해보세요
연관 상품을 찾고 있습니다...
Zenogen Pharma HSC-BANKER® GMP grade
For inquiries including COA (certificate of analysis), please send a message to:
zenogen_pharma-post@zenoaq.jp
Description
15 mL
For Cryopreservation of Hematopoietic Stem Cells
HSC-BANKER® GMP grade
ZENOAQ RESOURCE CO., LTD.
1-1 Shinmori Sakaegawa, Asaka-machi, Koriyama, Fukushima 963-0196 Japan
HSC-BANKER® GMP grade is an optimized cryopreservation medium for hematopoietic stem cells.
It is a ready-to-use product and has been reported that the cryopreservation outcomes of HSC-BANKER® are at least equivalent to those of the conventional protocol using DMSO and DEXTRAN.
It is completely free of serum and animal-derived components, and contains only USP, EP, JP graded ingredients or JP excipients.
Manufactured in a facility compliant with JP, EU, US, and PIC/S GMP guidelines, and quality is controlled under JP GMP standards.
※ This product has been registered in MF within PMDA in 2019.
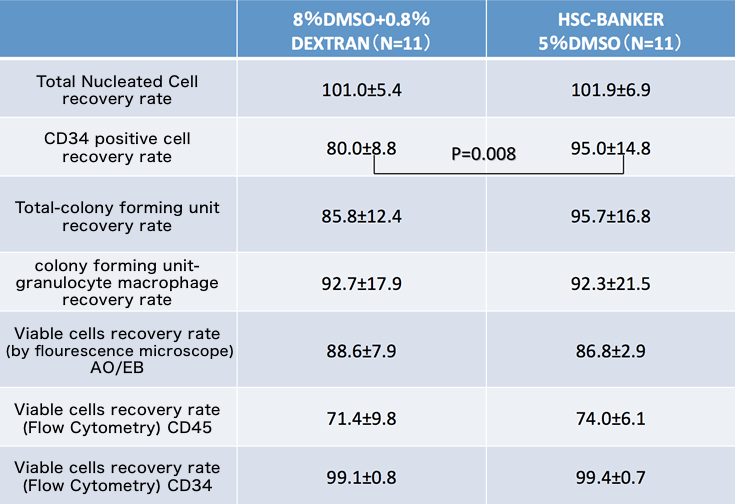
Comparative Study
This data is provided by Japan Red Cross Society.
| 항목 | 8%DMSO+0.8% DEXTRAN (N=11) | HSC-BANKER 5%DMSO (N=11) |
|---|---|---|
| Total Nucleated Cell recovery rate | 101.0±5.4 | 101.9±6.9 |
| CD34 positive cell recovery rate | 80.0±8.8 | 95.0±14.8 |
| P=0.008 | ||
| Total-colony forming unit recovery rate | 85.8±12.4 | 95.7±16.8 |
| colony forming unit-granulocyte macrophage recovery rate | 92.7±17.9 | 92.3±21.5 |
| Viable cells recovery rate (by fluorescence microscope) AO/EB | 88.6±7.9 | 86.8±2.9 |
| Viable cells recovery rate (Flow Cytometry) CD45 | 71.4±9.8 | 74.0±6.1 |
| Viable cells recovery rate (Flow Cytometry) CD34 | 99.1±0.8 | 99.4±0.7 |
Source: The Japan Society for Hematopoietic Cell Transplantation (2015)
Storage and Stability
| 항목 | 내용 |
|---|---|
| 저장 온도 | 2–8℃ |
| 안정성 | 제조일로부터 3년 (미개봉 상태, 지정 온도 보관 시) |
Freezing Protocol
- Remove red blood cells from the cord blood collection.
- Separate the cord blood into plasma and buffy coat fractions by centrifugation at 400 × g for 10 minutes.
- Reduce the volume of the cord blood to 13 mL by removing the plasma.
- Add gently an equal volume (13 mL) of HSC-BANKER® GMP grade to the cord blood, and place the freezing bag in a controlled-rate freezer to gradually freeze the cord blood to -80℃.
- After reaching below -80℃, transfer to a liquid nitrogen tank for long-term storage.
SDS
Disclaimer
HSC-BANKER® GMP grade is not itself a pharmaceutical.
ZENOAQ and Zenogen Pharma make no warranty, express or implied, as to the fitness or suitability of this product for any particular purpose and/or merchantability unless the use is intended for research.
제품 이미지
🏷️Zenogen Pharma 상품 둘러보기
동일 브랜드의 다른 상품들을 확인해보세요
배송/결제/교환/반품 안내
배송 정보
| 기본 배송비 |
| 교환/반품 배송비 |
|
|---|---|---|---|
| 착불 배송비 |
| ||
| 교환/반품 배송비 |
| ||
결제 및 환불 안내
| 결제수단 |
|
|---|---|
| 취소 |
|
| 반품 |
|
| 환급 |
|
교환 및 반품 접수
| 교환 및 반품 접수 기한 |
|
|---|---|
| 교환 및 반품 접수가 가능한 경우 |
|
| 교환 및 반품 접수가 불가능한 경우 |
|
교환 및 반품 신청
| 교환 절차 |
|
|---|---|
| 반품 절차 |
|